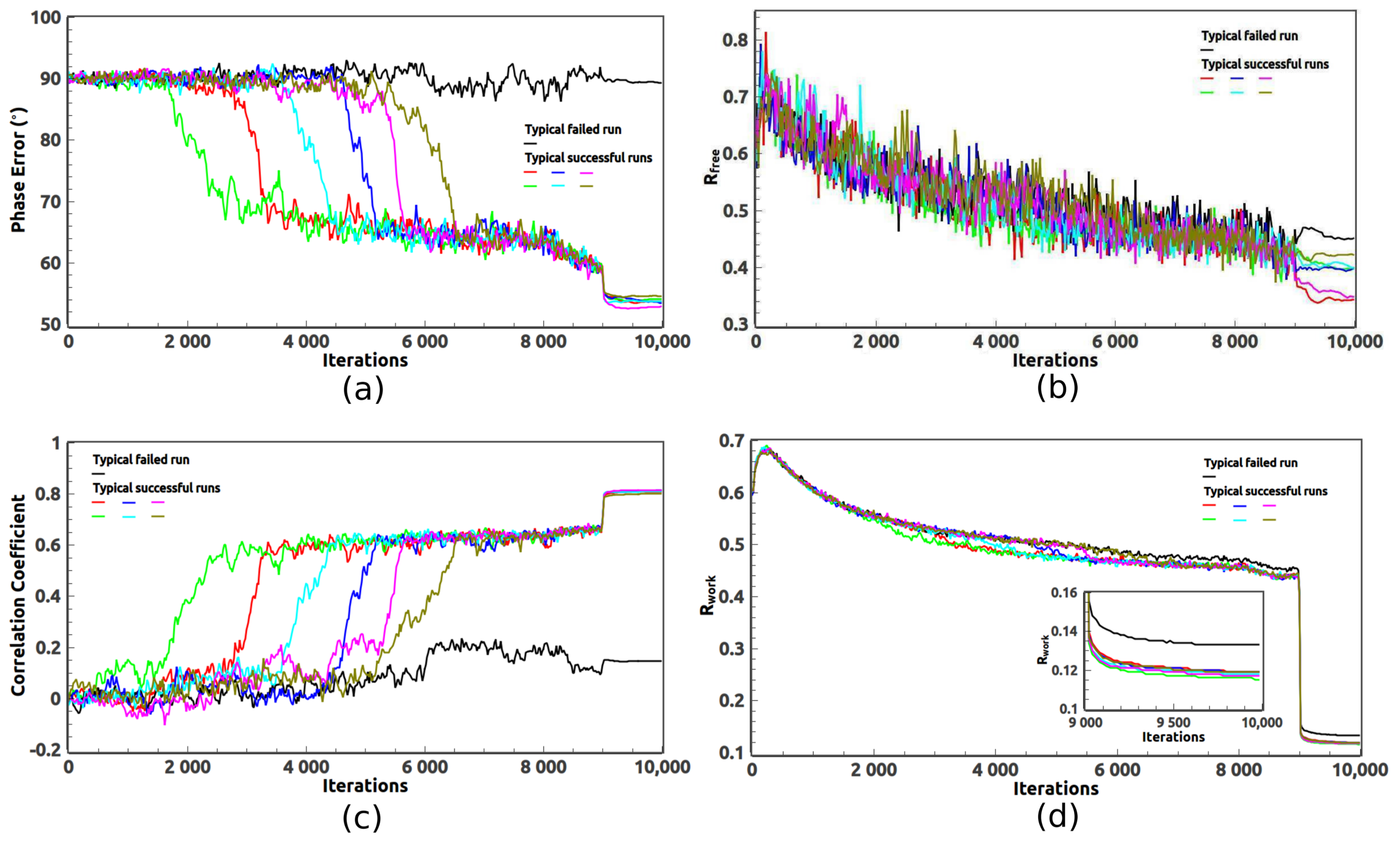
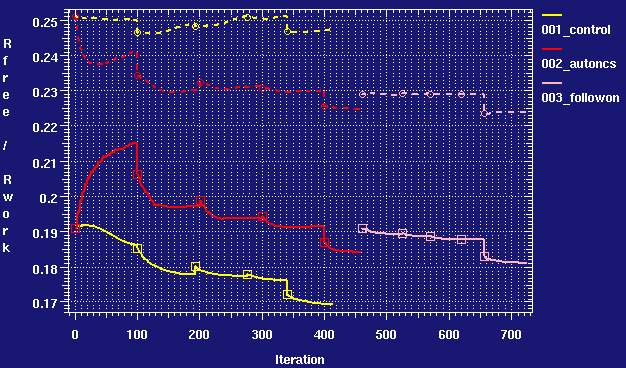
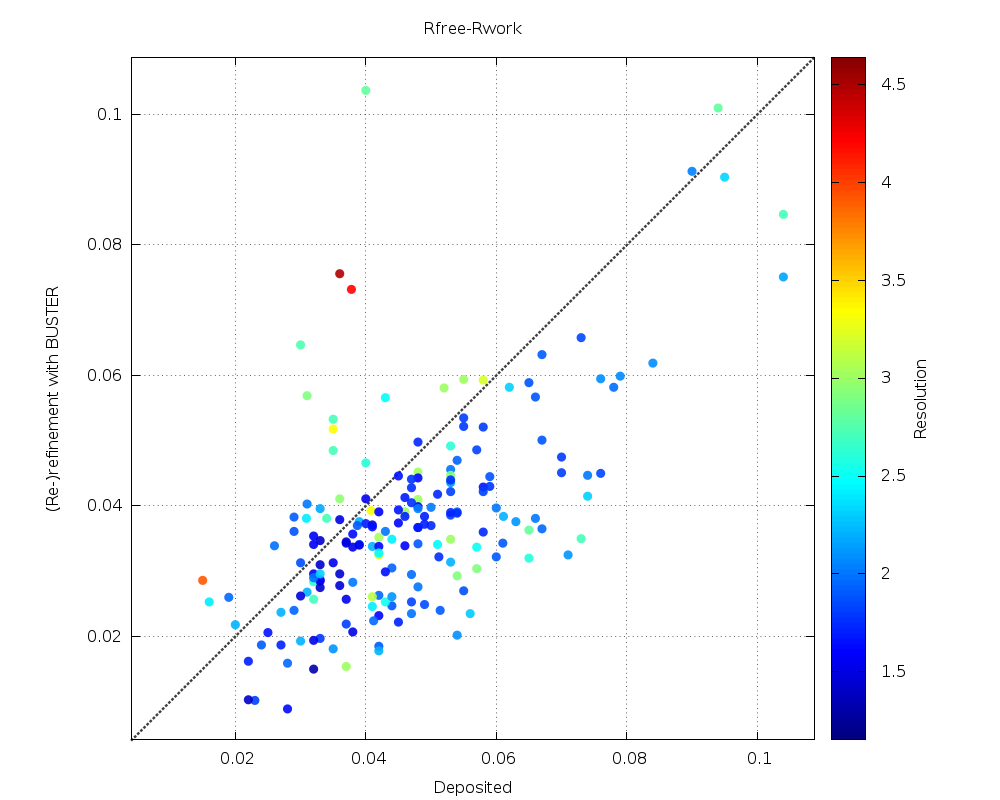
X-ray crystallography–based structural elucidation of enzyme-bound intermediates along the 1‑deoxy‑d‑xylulose 5-phosphat

A description of the structural determination procedures of a gap junction channel at 3.5 Å resolution – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free

Ultrasonic acoustic levitation for fast frame rate X-ray protein crystallography at room temperature | Scientific Reports
Use of knowledge-based restraints in phenix.refine to improve macromolecular refinement at low resolution

Tristan Croll on Twitter: "Some more fun with ISOLDE's adaptive restraints (testing for stability before releasing the official 1.0b5, as much as anything). Today I picked on 1z0v, a 3A crystal from

Tristan Croll on Twitter: "Some more fun with ISOLDE's adaptive restraints (testing for stability before releasing the official 1.0b5, as much as anything). Today I picked on 1z0v, a 3A crystal from

Tristan Croll on Twitter: "Well... remember what refinement is doing. Essentially, it's trying to make your model look as much like your data as it can, subject to the restraints you provide.

Macromolecular refinement of X-ray and cryo-electron microscopy structures with Phenix / OPLS3e for improved structure and ligand quality | bioRxiv