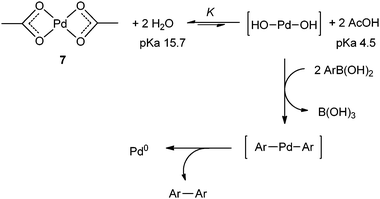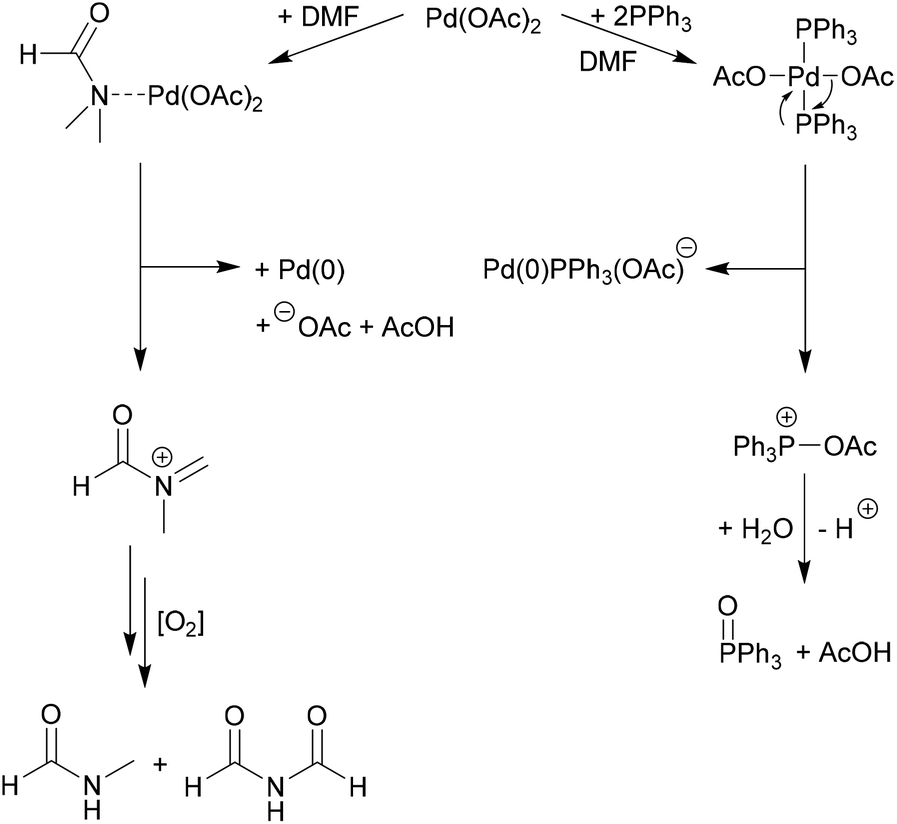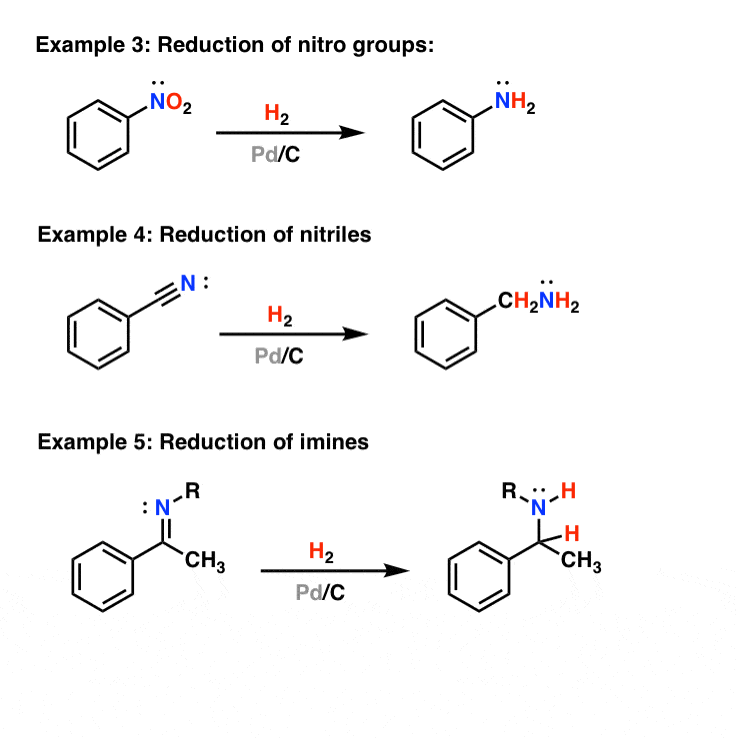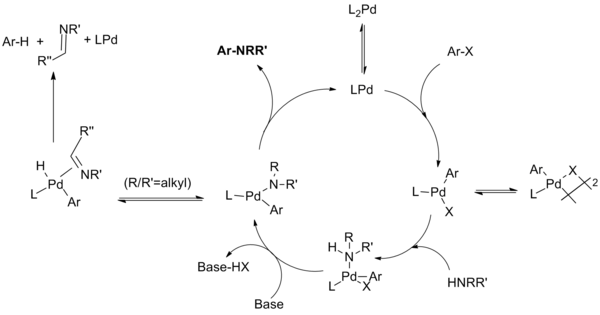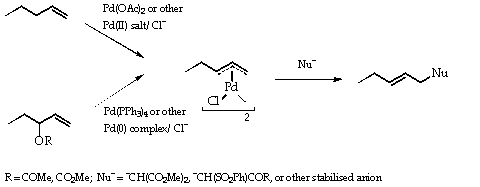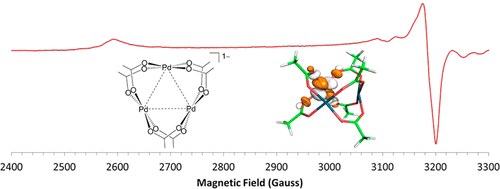
Palladium Acetate Revisited: Unusual Ring-Current Effects, One-Electron Reduction, and Metal–Metal Bonding - Inorg. Chem. - X-MOL

EP0921212A1 - Composition for electroplating palladium alloys and electroplating process using that composition - Google Patents
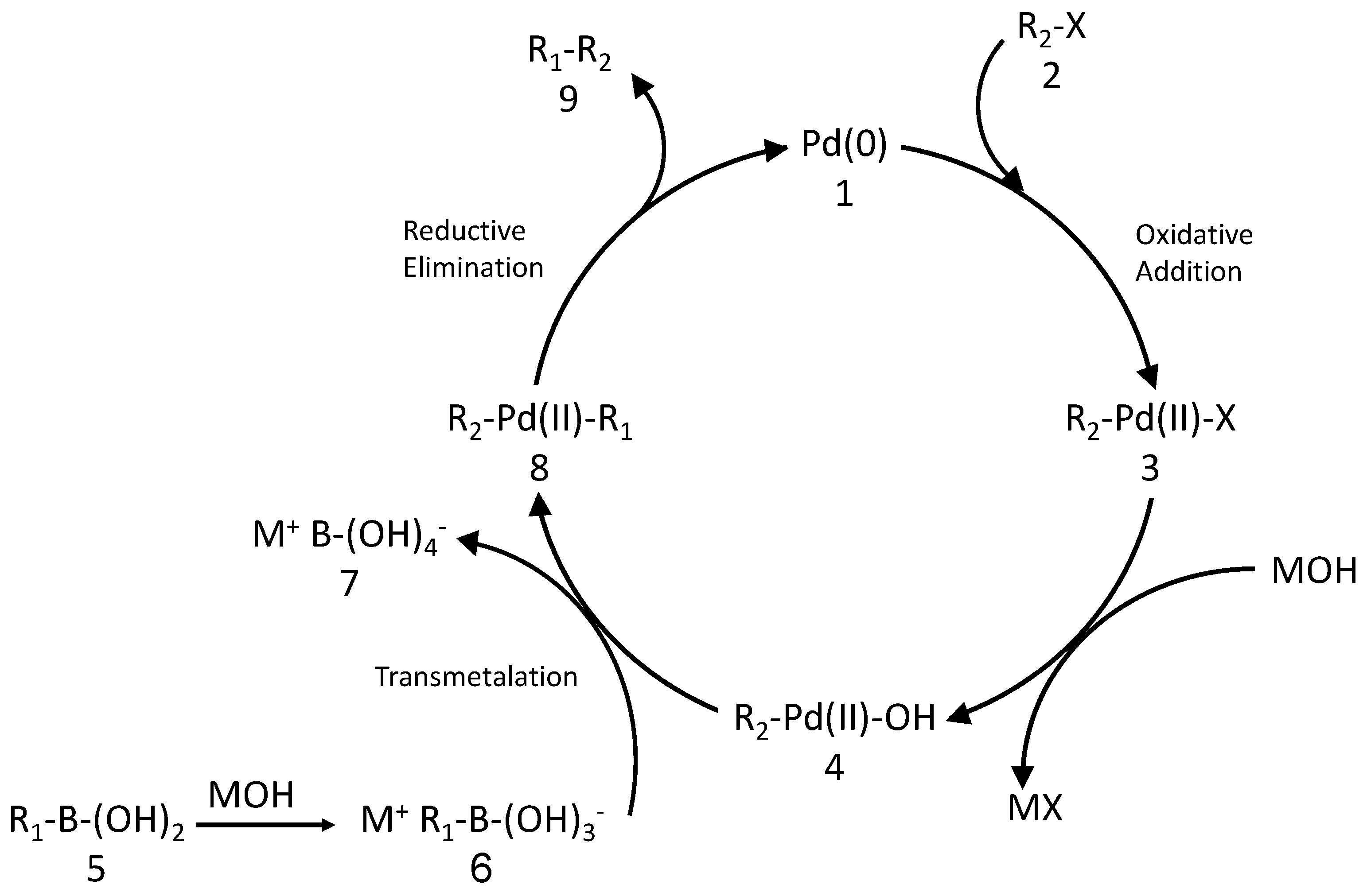
Catalysts | Free Full-Text | Recent Developments in the Immobilization of Palladium Complexes on Renewable Polysaccharides for Suzuki–Miyaura Cross-Coupling of Halobenzenes and Phenylboronic Acids | HTML
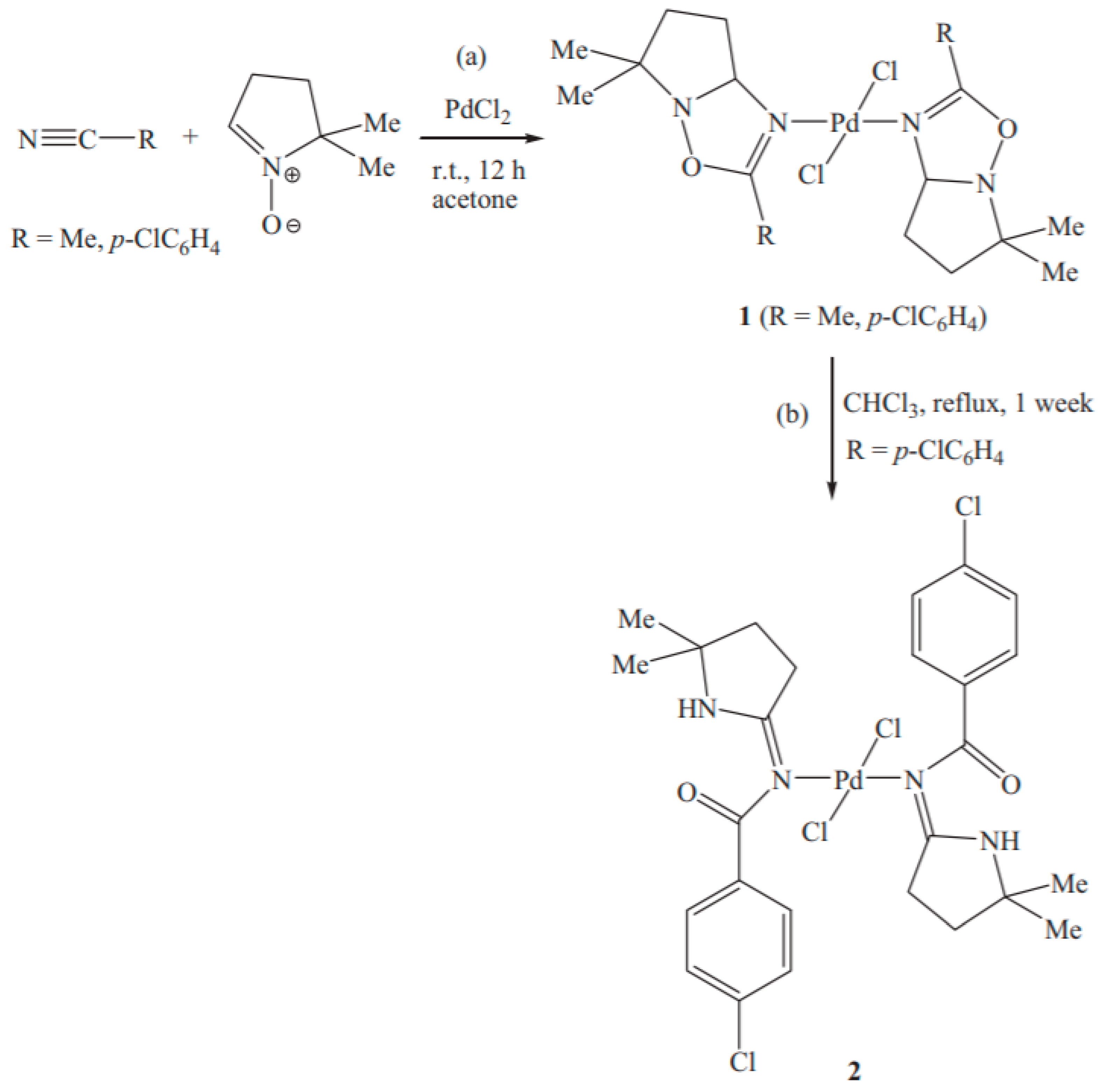
Catalysts | Free Full-Text | Recent Developments in the Immobilization of Palladium Complexes on Renewable Polysaccharides for Suzuki–Miyaura Cross-Coupling of Halobenzenes and Phenylboronic Acids | HTML
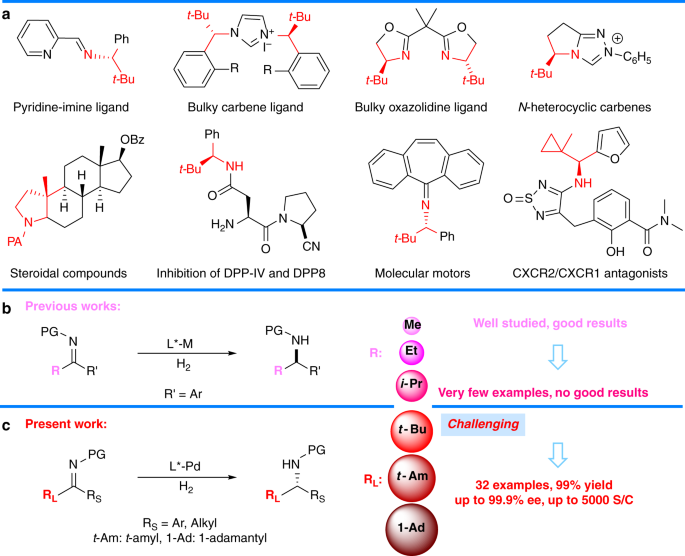
Pd(OAc) 2 -catalyzed asymmetric hydrogenation of sterically hindered N -tosylimines | Nature Communications
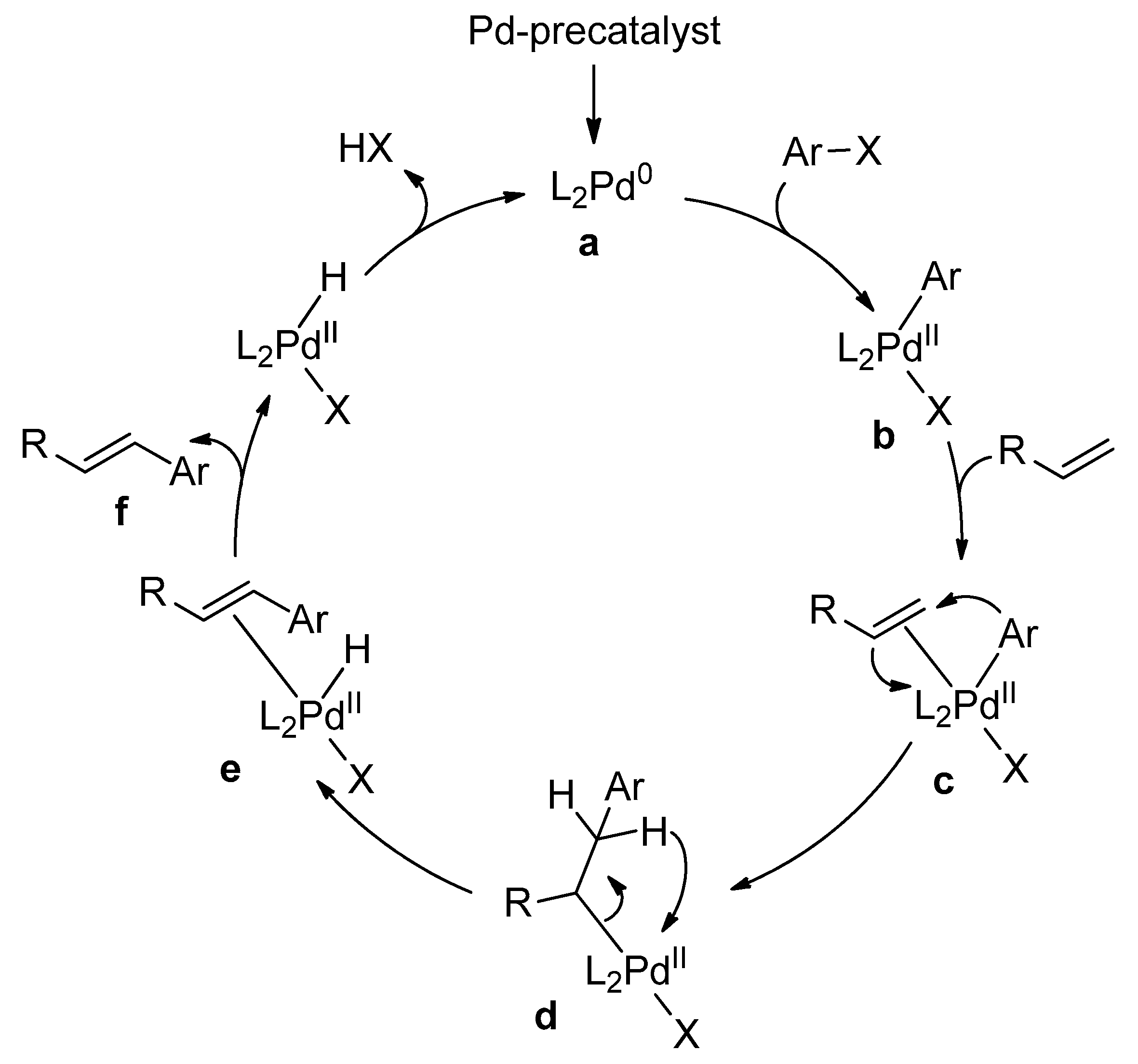
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML