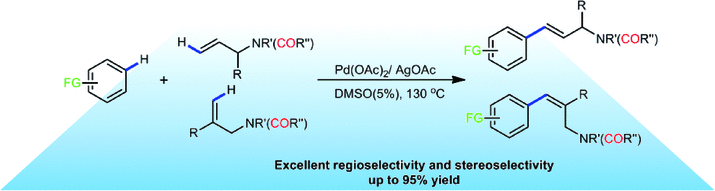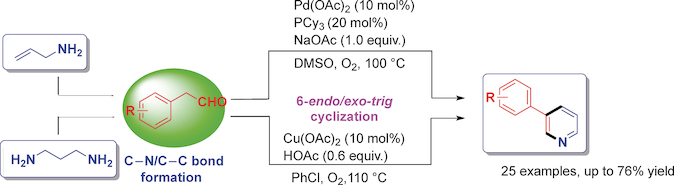
Synthesis of 3-Arylpyridines via Palladium/Copper-Catalyzed Annulation of Allylamine/1,3-Propanediamine and Aldehydes - Adv. Synth. Catal. - X-MOL

A steric tethering approach enables palladium-catalysed C–H activation of primary amino alcohols | Nature Chemistry
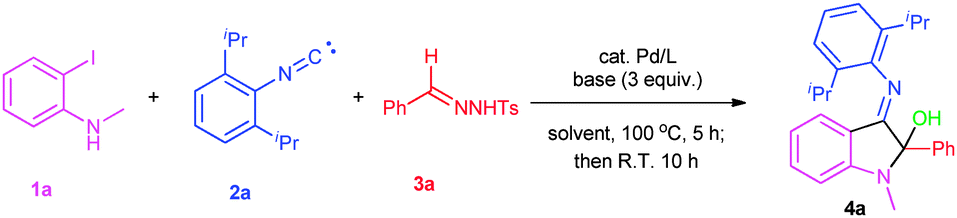
3-Aza π-allyl palladium derived from imino migration in palladium-carbene: MCRs toward multiple substituted indole skeleton - Chemical Communications (RSC Publishing) DOI:10.1039/C5CC05984D

Palladium(II)-Catalyzed C–H Activation and C–C Coupling/Cyclization of Benzamidine and Terminal Alkynes Using an Internal Oxidant - Synlett - X-MOL

Palladium(II)‐Catalyzed Sequential Aminopalladation and Oxidative Coupling with Acetylenes/Enones: Synthesis of Newly Substituted Quinolines from 2‐Aminophenyl Propargyl Alcohols - Thirupathi - 2016 - Advanced Synthesis & Catalysis - Wiley Online ...
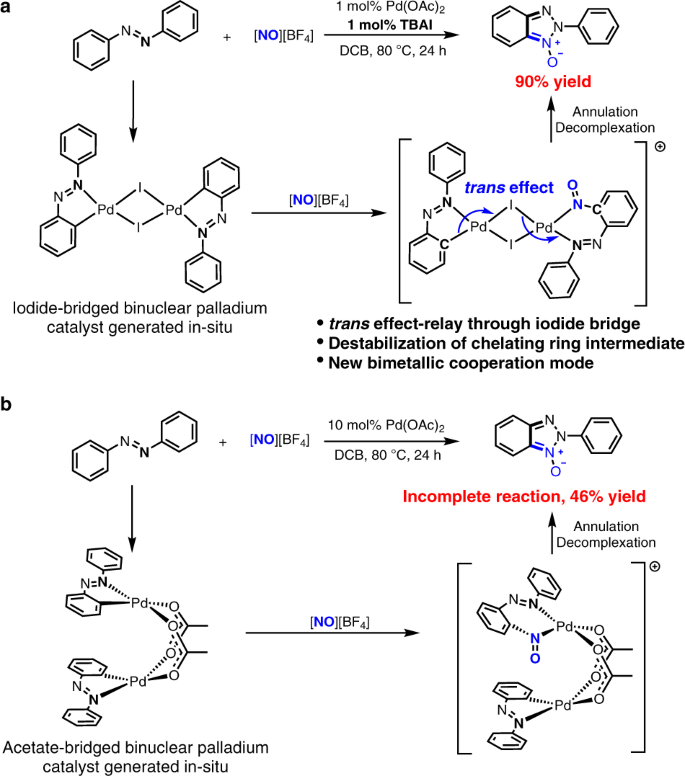
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry

Palladium‐Catalyzed Regio‐ and Chemoselective Reactions of 2‐Bromobenzyl Bromides: Expanding the Scope for the Synthesis of Biaryls Fused to a Seven‐Membered Sultam - Laha - 2015 - European Journal of Organic Chemistry -
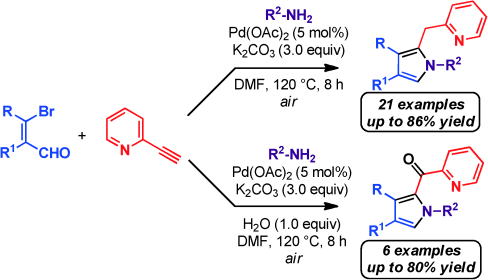
Palladium‐Catalyzed One‐Pot Sonogashira Coupling, exo‐dig Cyclization and Hydride Transfer Reaction: Synthesis of Pyridine‐Substituted Pyrroles,Advanced Synthesis & Catalysis - X-MOL
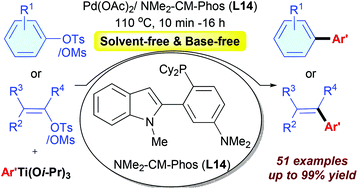
Palladium-catalyzed cross-coupling of (hetero)aryl or alkenyl sulfonates with aryl titanium as the multi-functional reagent - Organic Chemistry Frontiers (RSC Publishing)

The mechanism of palladium(II)-mediated C–H cleavage with mono-N-protected amino acid (MPAA) ligands: origins of rate acceleration in: Pure and Applied Chemistry Volume 88 Issue 1-2 (2016)
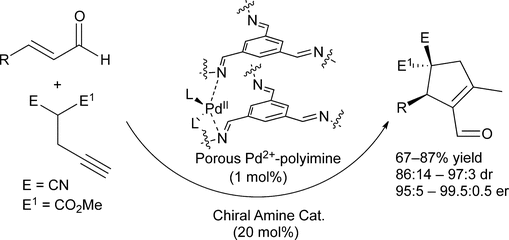
The Use of Porous Palladium(II)‐polyimine in Cooperatively‐ catalyzed Highly Enantioselective Cascade Transformations - Adv. Synth. Catal. - X-MOL

Safe Removal of the Allyl Protecting Groups of Allyl Esters using a Recyclable, Low‐Leaching and Ligand‐Free Palladium Nanoparticle Catalyst - Takagi - 2015 - Advanced Synthesis & Catalysis - Wiley Online Library
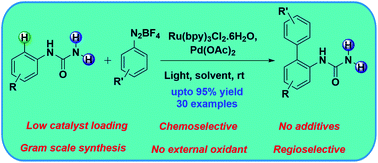
Dual palladium–photoredox catalyzed chemoselective C–H arylation of phenylureas - Chemical Communications (RSC Publishing)
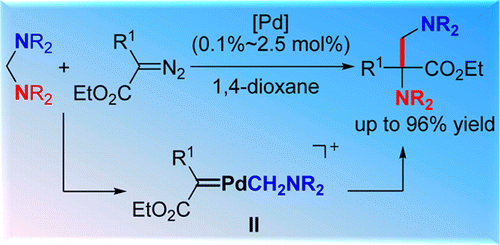
Palladium-Catalyzed Formal Insertion of Carbenoids into Aminals via C–N Bond Activation----lanzhou Branch;Chinese Academy of Sciences