
Cross-Coupling and Dehalogenation Reactions Catalyzed by (N-Heterocyclic carbene)Pd(allyl)Cl Complexes

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Palladium catalyzed-dehalogenation of aryl chlorides and bromides using phosphite ligands - ScienceDirect

Palladium catalyzed-dehalogenation of aryl chlorides and bromides using phosphite ligands - ScienceDirect

Pd/C-catalyzed room-temperature hydrodehalogenation of aryl halides with hydrazine hydrochloride - ScienceDirect
![PDF] Reactivity of Aryl Halides for Reductive Dehalogenation in (Sea)water Using Polymer-Supported Terpyridine Palladium Catalyst | Semantic Scholar PDF] Reactivity of Aryl Halides for Reductive Dehalogenation in (Sea)water Using Polymer-Supported Terpyridine Palladium Catalyst | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/faba9f24ba08cf7fb7c9ca0b5427112c22e9fa41/3-Table1-1.png)
PDF] Reactivity of Aryl Halides for Reductive Dehalogenation in (Sea)water Using Polymer-Supported Terpyridine Palladium Catalyst | Semantic Scholar
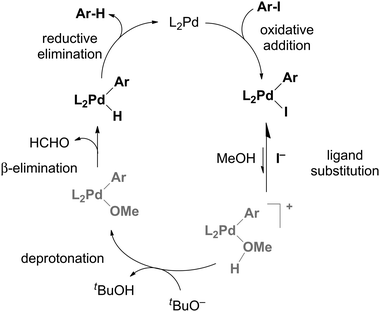
A mechanistic investigation of hydrodehalogenation using ESI-MS - Chemical Communications (RSC Publishing) DOI:10.1039/C3CC46271D

Molecules | Free Full-Text | Reactivity of Aryl Halides for Reductive Dehalogenation in (Sea)water Using Polymer-Supported Terpyridine Palladium Catalyst

Metal catalyzed defunctionalization reactions - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB01949D
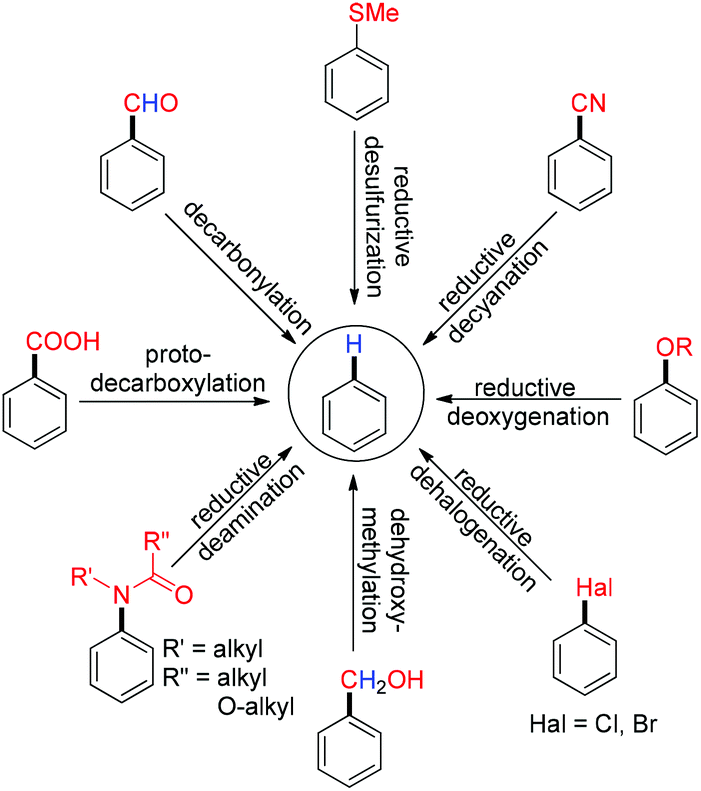
Metal catalyzed defunctionalization reactions - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB01949D
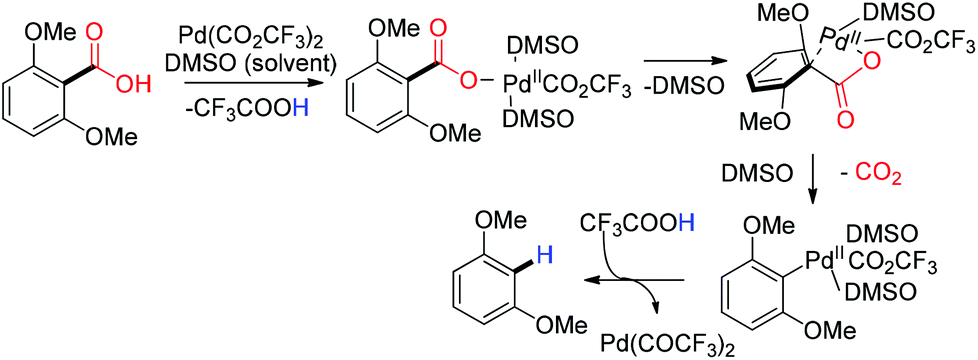
Metal catalyzed defunctionalization reactions - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB01949D
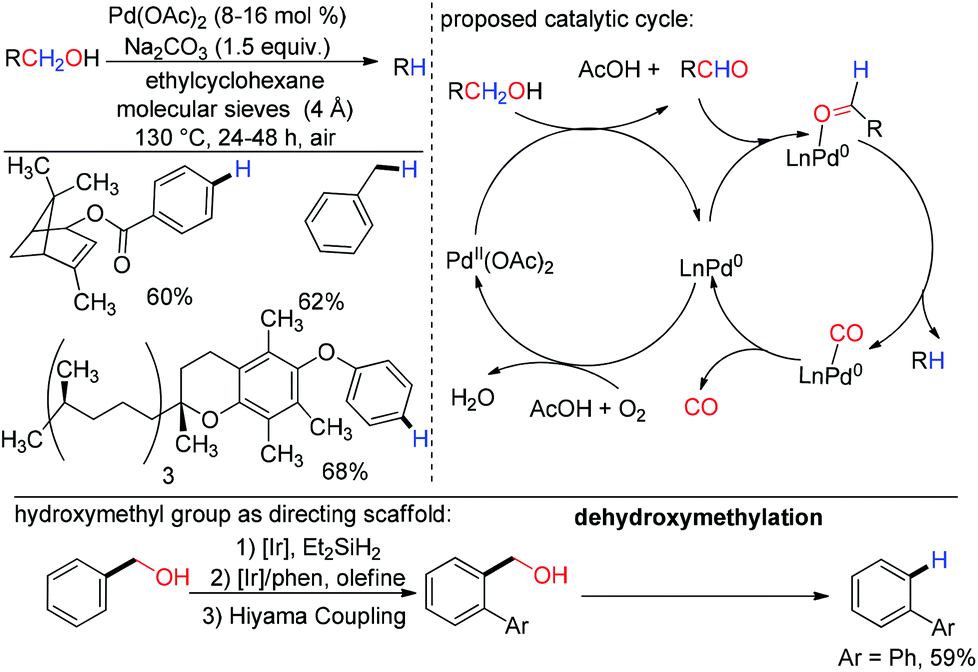
Metal catalyzed defunctionalization reactions - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB01949D

Diphenylphosphinite ionic liquid (IL-OPPh2): A solvent and ligand for palladium-catalyzed silylation and dehalogenation reaction of aryl halides with triethylsilane - ScienceDirect

Cover Picture: Development of Palladium Surface‐Enriched Heteronuclear Au–Pd Nanoparticle Dehalogenation Catalysts in an Ionic Liquid (Chem. Eur. J. 4/2013) - Yuan - 2013 - Chemistry – A European Journal - Wiley Online Library

Molecules | Free Full-Text | Reactivity of Aryl Halides for Reductive Dehalogenation in (Sea)water Using Polymer-Supported Terpyridine Palladium Catalyst

Ligand-Free, Palladium-Catalyzed Dihydrogen Generation from TMDS: Dehalogenation of Aryl Halides on Water
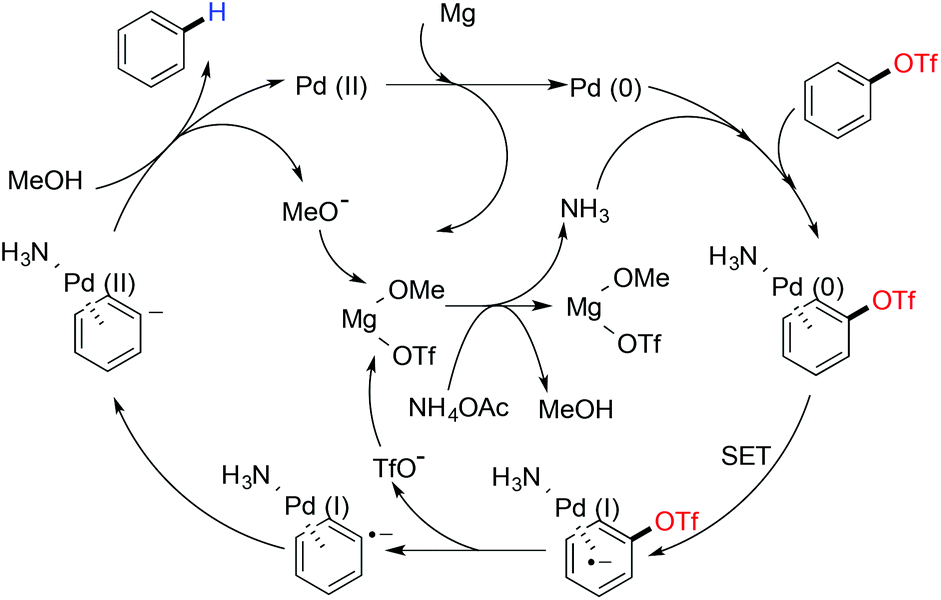
Metal catalyzed defunctionalization reactions - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB01949D
![PDF) Synthesis of the novel ionic liquid [N-pentylpyridinium]+ [closo-CB11H12]? and its usage as a reaction medium in catalytic dehalogenation of aromatic halides | Keith Carpenter and Zhu Yinghuai - Academia.edu PDF) Synthesis of the novel ionic liquid [N-pentylpyridinium]+ [closo-CB11H12]? and its usage as a reaction medium in catalytic dehalogenation of aromatic halides | Keith Carpenter and Zhu Yinghuai - Academia.edu](https://0.academia-photos.com/attachment_thumbnails/44287312/mini_magick20190214-14059-egfzk1.png?1550201499)
PDF) Synthesis of the novel ionic liquid [N-pentylpyridinium]+ [closo-CB11H12]? and its usage as a reaction medium in catalytic dehalogenation of aromatic halides | Keith Carpenter and Zhu Yinghuai - Academia.edu

A practical and highly efficient reductive dehalogenation of aryl halides using heterogeneous Pd/AlO(OH) nanoparticles and sodium borohydride - Tetrahedron - X-MOL
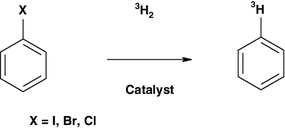
Scheme 1 | Understanding the observed decreasing specific activity value in the series: aryl iodide, aryl bromide, aryl chloride during palladium catalyzed tritium dehalogenations | SpringerLink

![2 Plausible Pd-catalyzed Dehalogenation Mechanism [40, 45] | Download Scientific Diagram 2 Plausible Pd-catalyzed Dehalogenation Mechanism [40, 45] | Download Scientific Diagram](https://www.researchgate.net/profile/Milandip_Karak/publication/322568978/figure/fig27/AS:614379531825192@1523490903688/Plausible-Pd-catalyzed-Dehalogenation-Mechanism-40-45.png)



