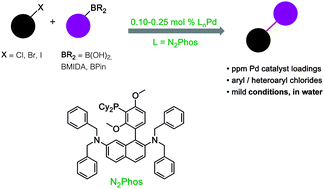
Heterogeneous versus Homogeneous Palladium Catalysts for Cross‐Coupling Reactions - Pagliaro - 2012 - ChemCatChem - Wiley Online Library
![Room-Temperature, Copper-Free Sonogashira Reactions Facilitated by Air-Stable, Monoligated Precatalyst [DTBNpP] Pd(crotyl)Cl. - Abstract - Europe PMC Room-Temperature, Copper-Free Sonogashira Reactions Facilitated by Air-Stable, Monoligated Precatalyst [DTBNpP] Pd(crotyl)Cl. - Abstract - Europe PMC](https://europepmc.org/articles/PMC6644404/bin/ao-2018-01868w_0009.jpg)
Room-Temperature, Copper-Free Sonogashira Reactions Facilitated by Air-Stable, Monoligated Precatalyst [DTBNpP] Pd(crotyl)Cl. - Abstract - Europe PMC
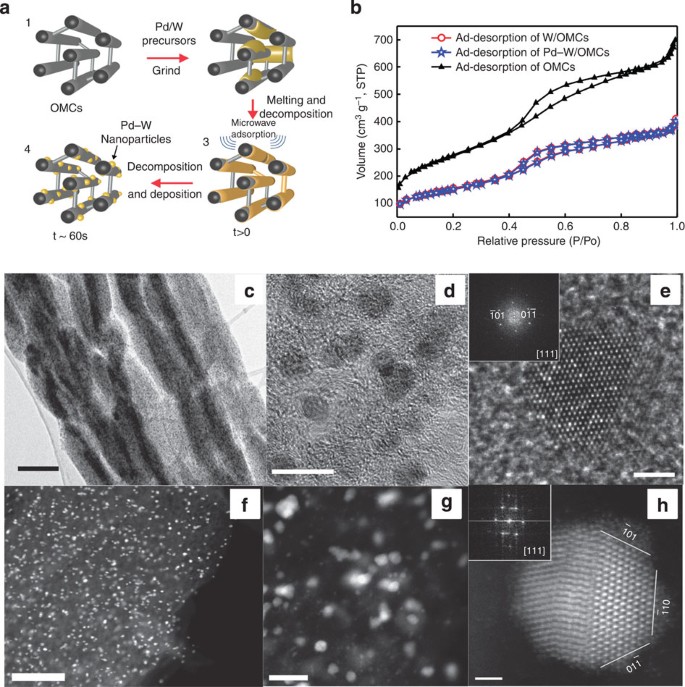
Small palladium islands embedded in palladium–tungsten bimetallic nanoparticles form catalytic hotspots for oxygen reduction | Nature Communications

β-Diketimine appended periodic mesoporous organosilica as a scaffold for immobilization of palladium acetate: An efficient green catalyst for Wacker type reaction - ScienceDirect

EP2116574A1 - Composition containing pyrimidine compound and luminescent element employing the composition - Google Patents

Table 1 from Palladium(II) complexes with highly basic imidazolin-2-imines and their reactivity toward small bio-molecules. | Semantic Scholar
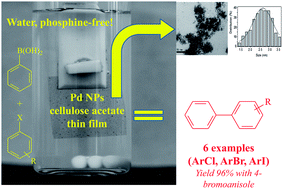
Palladium nanoparticles supported in a polymeric membrane: an efficient phosphine-free “green” catalyst for Suzuki–Miyaura reactions in water - RSC Advances (RSC Publishing)
Supported Palladium Nanoparticles Synthesized by Living Plants as a Catalyst for Suzuki-Miyaura Reactions
Supported Palladium Nanoparticles Synthesized by Living Plants as a Catalyst for Suzuki-Miyaura Reactions
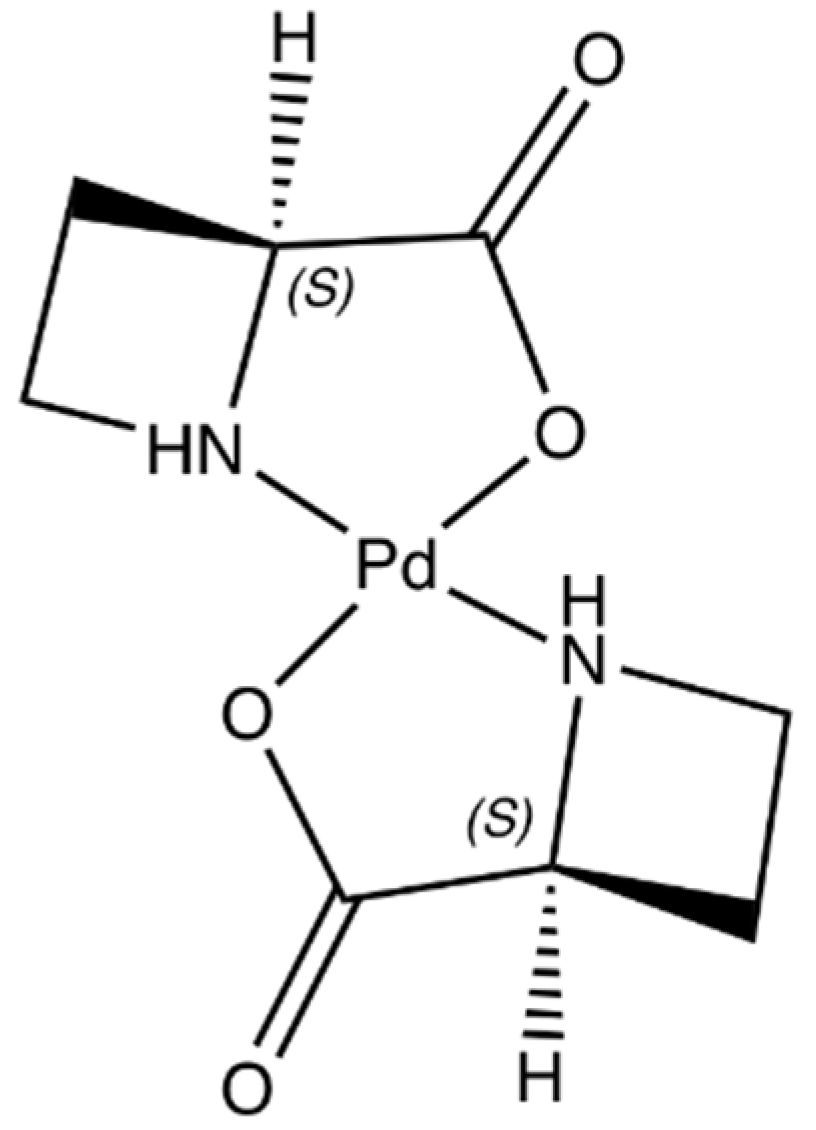
Catalysts | Free Full-Text | Synthesis, Structure, and Catalytic Reactivity of Pd(II) Complexes of Proline and Proline Homologs | HTML

Ligand and Solvent Selection for Enhanced Separation of Palladium Catalysts by Organic Solvent Nanofiltration. - Abstract - Europe PMC

PDF) Palladium complexes containing imino phenoxide ligands: synthesis, luminescence, and their use as catalysts for the ring-opening polymerization of rac-lactide
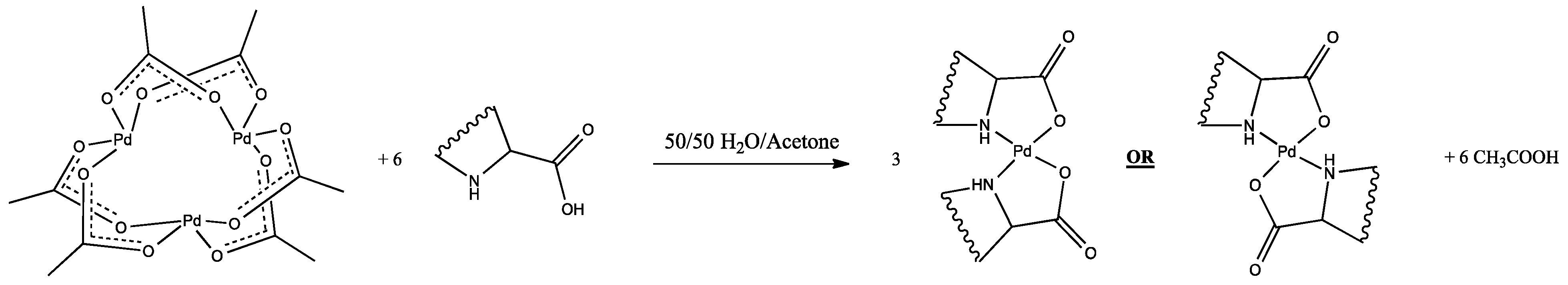
Catalysts | Free Full-Text | Synthesis, Structure, and Catalytic Reactivity of Pd(II) Complexes of Proline and Proline Homologs | HTML

PDF) The Effect of Bases, Catalyst Loading and Reaction Temperature on the Catalytic Evaluation of Supported Palladium(II) Catalyst in the Mizoroki-Heck

Regioselective cyclometallation of N-methyl-N-(naphthalen-2-ylmethyl)-2-(pyridin-2-yl)ethanamine with palladium(II) acetate and catalytic reduction of various functional groups - ScienceDirect
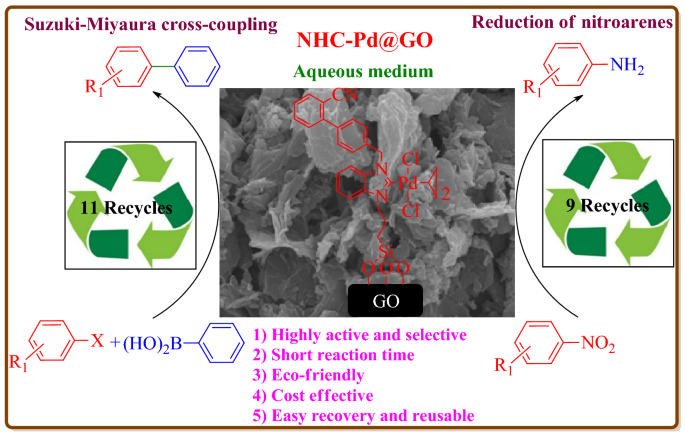
Immobilized N -Heterocyclic Carbene-Palladium(II) Complex on Graphene Oxide as Efficient and Recyclable Catalyst for Suzuki–Miyaura Cross-Coupling and Reduction of Nitroarenes | SpringerLink
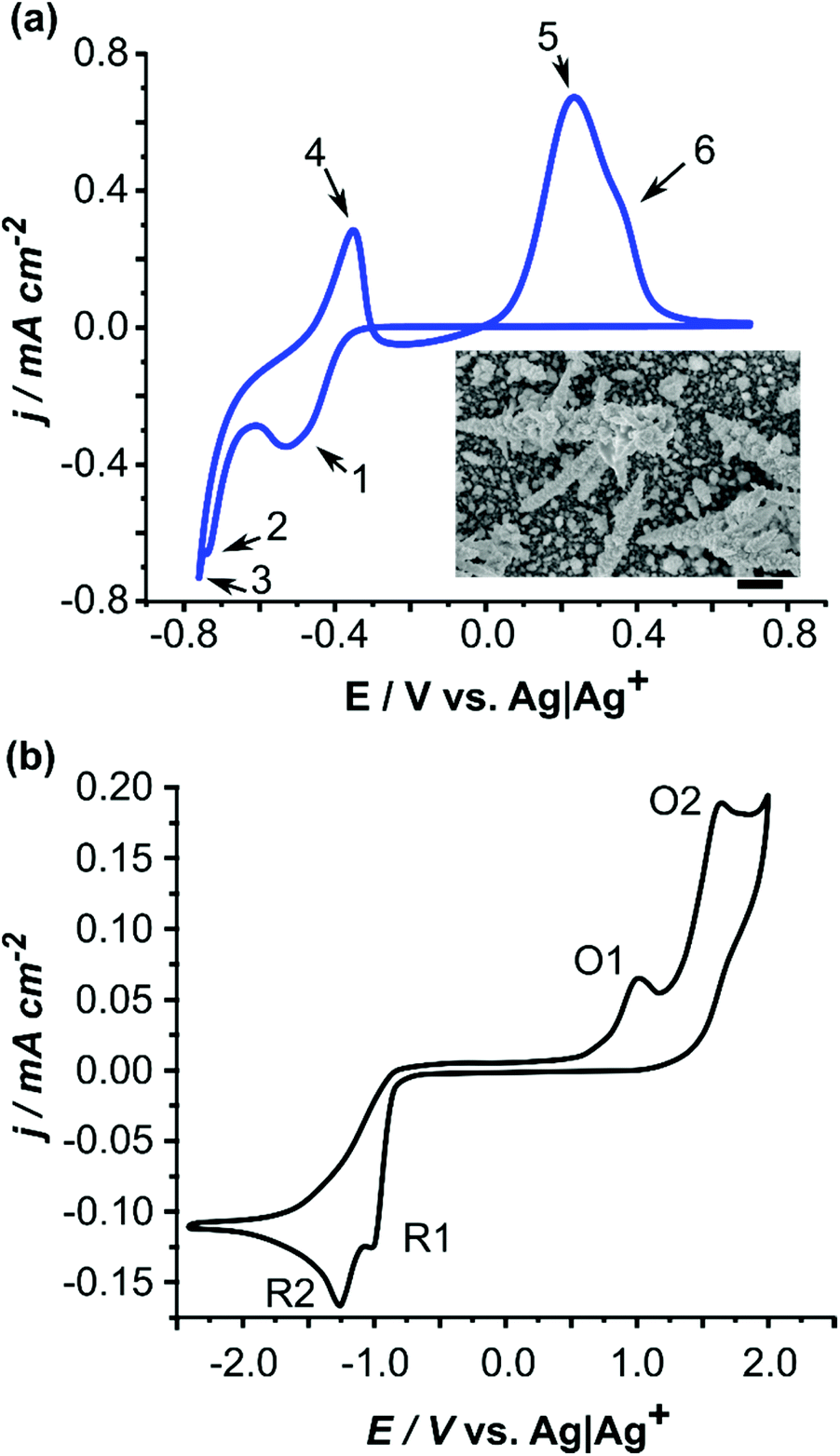
Switching on palladium catalyst electrochemical removal from a palladium acetate–acetonitrile system via trace water addition - Green Chemistry (RSC Publishing)