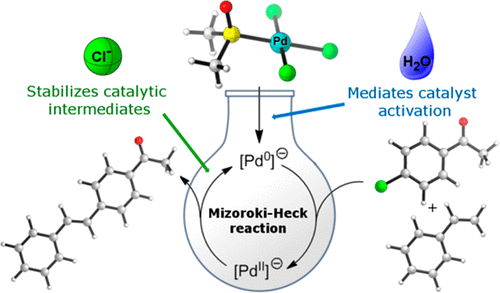
Dimethyl Sulfoxide: Yesterday's Solvent, Today's Reagent - Tashrifi - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library

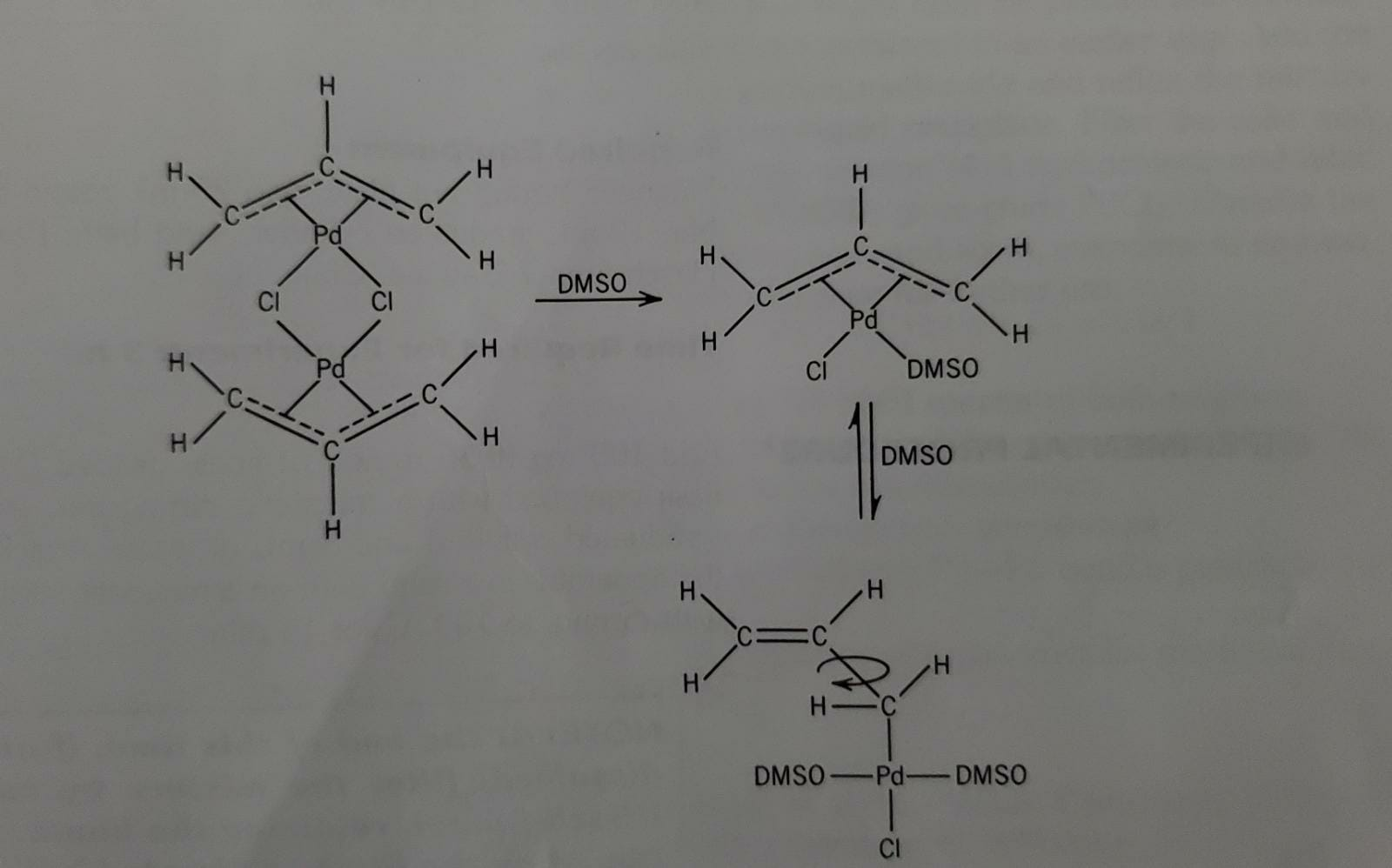
Crystal structure of (acridine)dichloro(dimethyl sulfoxide-)S)- palladium (II)—dimethyl sulfoxide (1:2), PdCl2(C13H9N)(C2H6SO) · 2C2H6SO : Zeitschrift für Kristallographie - New Crystal Structures

Figure 6 from Dimethyl Sulfoxide Containing Platinum(II) and Palladium(II) Chelate Complexes of Glyoxylic and Pyruvic Acid Thiosemicarbazones. A New Class of Cytotoxic Metal Complexes - Semantic Scholar

Figure 2 from ESI-MS studies of palladium (II) complexes with 1-(p-toluenesulfonyl)cytosine/cytosinato ligands. | Semantic Scholar

Figure 1 from Kinetics and mechanism of palladium(II) catalysed oxidation of dimethyl sulfoxide by alkaline periodate | Semantic Scholar

New mono- and dinuclear complexes of 7-azaindole-3-carboxaldehyde with palladium(II): crystal structure, IR and Raman spectra, DFT calculations and in vitro antiproliferative activity - ScienceDirect
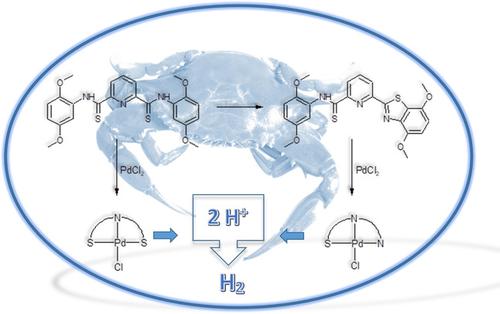
SNS versus SNN Pincer Ligands: Electrochemical Studies and Their Palladium(II) Complexes as Electro‐Catalyst for Proton Reduction,ChemistrySelect - X-MOL

Deposition of palladium nanoparticles on the silicon surface via galvanic replacement in DMSO | SpringerLink

Complexes of platinum and palladium with β-diketones and DMSO: Synthesis, characterization, molecular modeling, and biological studies - ScienceDirect
![Palladium(II)‐Catalyzed Direct Access to Indeno[1,2‐c]isochromen‐5(11H)‐Ones via Intramolecular Oxypalladation‐Initiated Cascade Process - Karuppasamy - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library Palladium(II)‐Catalyzed Direct Access to Indeno[1,2‐c]isochromen‐5(11H)‐Ones via Intramolecular Oxypalladation‐Initiated Cascade Process - Karuppasamy - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/991d9f07-8d3f-4784-ac78-1c5230a1bfc8/adsc202000456-toc-0001-m.jpg)
Palladium(II)‐Catalyzed Direct Access to Indeno[1,2‐c]isochromen‐5(11H)‐Ones via Intramolecular Oxypalladation‐Initiated Cascade Process - Karuppasamy - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library
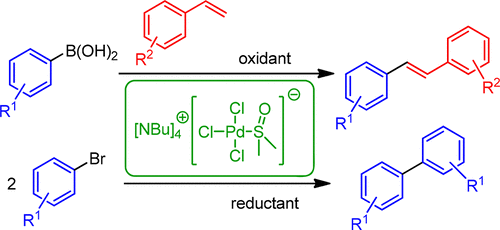
Oxidative and Reductive Cross-Coupling Reactions Catalyzed by an Anionic “Ligandless” Palladium Complex - Org. Process Res. Dev. - X-MOL
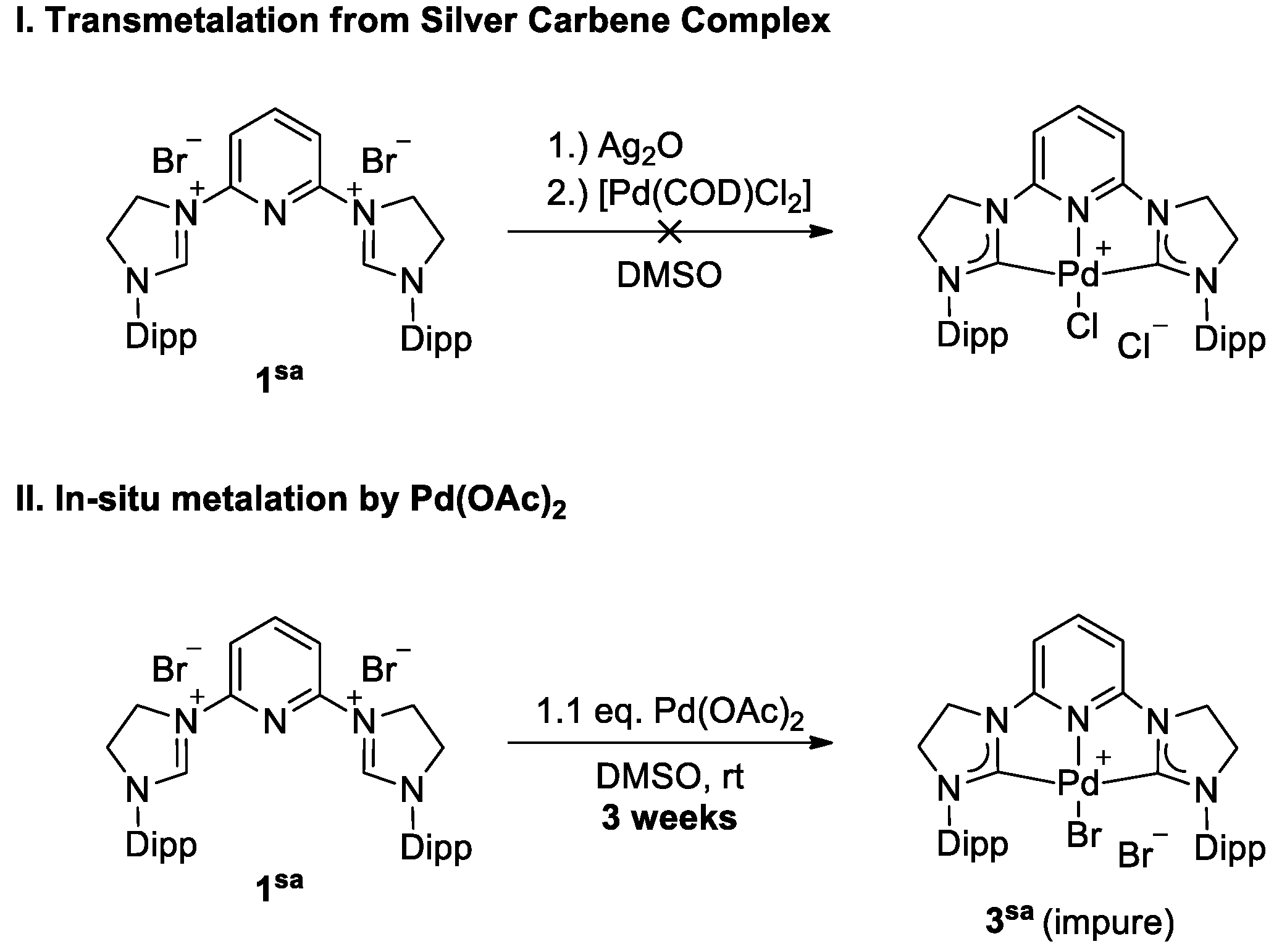
Inorganics | Free Full-Text | Transmetalation from Magnesium–NHCs—Convenient Synthesis of Chelating π-Acidic NHC Complexes | HTML

Influence of water/DMSO ratio on reaction rate in palladium-catalyzed... | Download Scientific Diagram
DMSO-mediated palladium-catalyzed cyclization of two isothiocyanates via C–H sulfurization: a new route to 2-aminobenzothiazol

Figure 1 from A palladium(II) complex containing both carbonyl and imine oxime ligands: crystal structure, experimental and theoretical UV-vis, IR and NMR studies. | Semantic Scholar

PDF) Synthesis and Identification of Pd(II) and Ni(II) Dithiooxamide Complexes- Part I: Experimental

Complexes of platinum and palladium with β-diketones and DMSO: Synthesis, characterization, molecular modeling, and biological studies - ScienceDirect
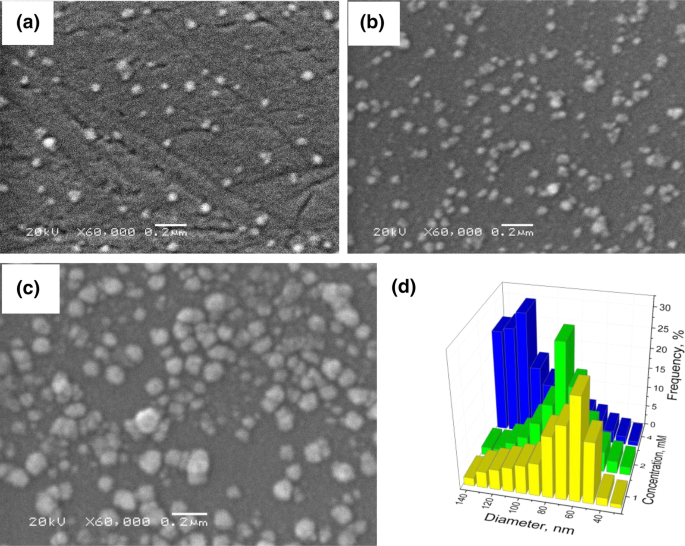
Deposition of palladium nanoparticles on the silicon surface via galvanic replacement in DMSO | SpringerLink
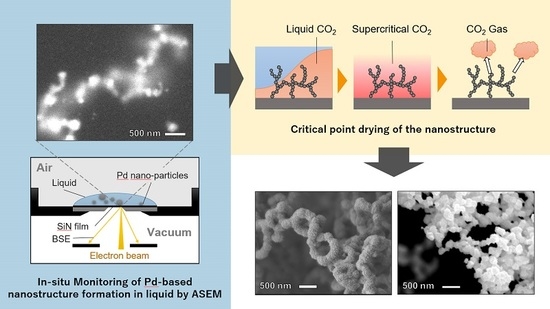
IJMS | Free Full-Text | Network of Palladium-Based Nanorings Synthesized by Liquid-Phase Reduction Using DMSO-H2O: In Situ Monitoring of Structure Formation and Drying Deformation by ASEM
Figure S2. 1 H NMR spectra in DMSO-d 6 of polymers functionalized with... | Download Scientific Diagram

Mild aromatic palladium-catalyzed protodecarboxylation: kinetic assessment of the decarboxylative palladation and the protodepalladation steps. - Abstract - Europe PMC