Stable Surfaces That Bind Too Tightly: Can Range-Separated Hybrids or DFT+U Improve Paradoxical Descriptions of Surface Chemistry? - Abstract - Europe PMC

Describing static correlation in bond dissociation by Kohn–Sham density functional theory: The Journal of Chemical Physics: Vol 122, No 9

Comparison of the performance of exact-exchange-based density functional methods: The Journal of Chemical Physics: Vol 137, No 11

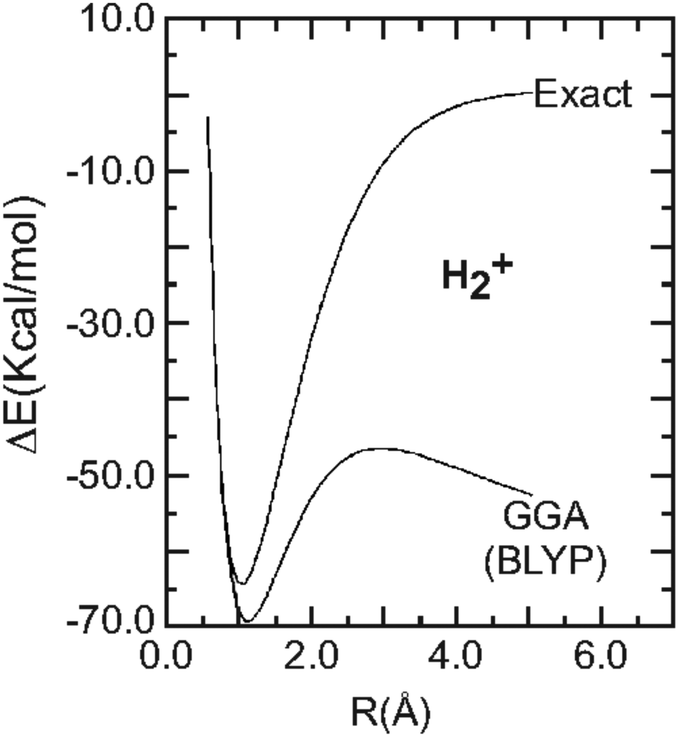
Density functionals that are one- and two- are not always many-electron self-interaction-free, as shown for H2+, He2+, LiH+, and Ne2+: The Journal of Chemical Physics: Vol 126, No 10

Pressure and temperature effects on water dissociation reaction for hydrogen production over zeolites - ScienceDirect

The role of dissociation channels of excited electronic states in quantum optimal control of ozone isomerization: A three-state dynamical model - ScienceDirect
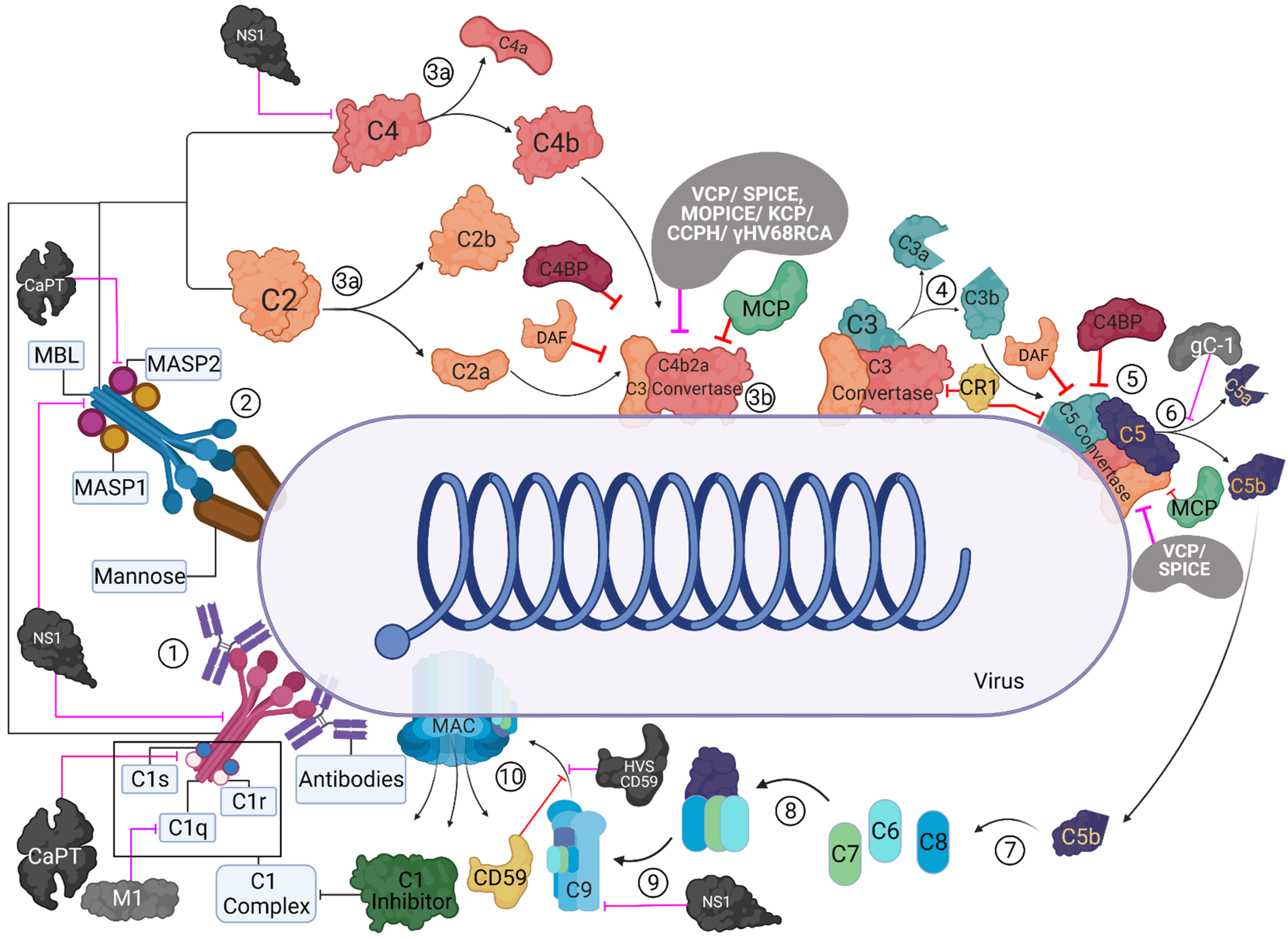
Viruses | Free Full-Text | Complement Proteins as Soluble Pattern Recognition Receptors for Pathogenic Viruses | HTML
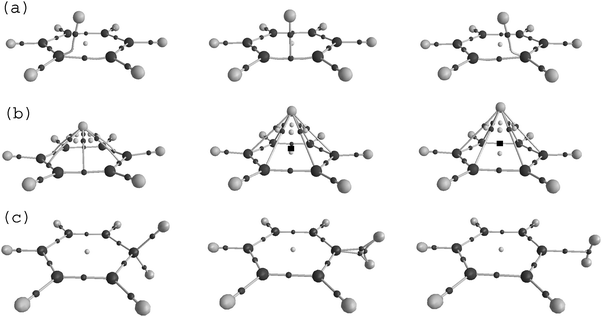
Energetic and electron density analysis of hydrogen dissociation of protonated benzene - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/B906961E

Two‐ and three‐state conical intersections in the electron capture dissociation of disulfides: The importance of multireference calculations - Gámez - 2011 - International Journal of Quantum Chemistry - Wiley Online Library

INSIGHTS INTO HYDROCARBON CHAIN AND AROMATIC RING FORMATION IN THE INTERSTELLAR MEDIUM: COMPUTATIONAL STUDY OF THE ISOMERS OF AND AND THEIR FORMATION PATHWAYS - IOPscience

Mechanistic insight into the photoredox catalysis of anti-markovnikov alkene hydrofunctionalization reactions. - Abstract - Europe PMC

Density functionals that are one- and two- are not always many-electron self-interaction-free, as shown for H2+, He2+, LiH+, and Ne2+: The Journal of Chemical Physics: Vol 126, No 10